How To Draw Microstructure From Phase Diagram
Alloy metals tin be in dissimilar phases. Phases are physically homogeneous states of an alloy. A phase has a precise chemical composition – a certain arrangement and bonding between the atoms.
This structure of atoms imparts unlike properties to different phases. Nosotros tin can choose the phase we want and use it in our applications.
Only some special alloys can exist in multiple phases. Heating the metal to specific temperatures using heat handling procedures results in different phases. Some special alloys can exist in more than i phase at the same temperature.
What Are Phase Diagrams?
Phase diagrams are graphical representations of the phases present in an alloy at different conditions of temperature, pressure, or chemical limerick.
The diagram describes the suitable conditions for ii or more than phases to exist in equilibrium. For instance, the water phase diagram describes a point (triple point) where water tin can coexist in three different phases at the same fourth dimension. This happens at just to a higher place the freezing temperature (0.01°C) and 0.006 atm.
Using the Diagrams
There are four major uses of blend phase diagrams:
- Development of new alloys based on awarding requirements.
- Production of these alloys.
- Development and control of advisable heat treatment procedures to improve the chemical, physical, and mechanical properties of these new alloys.
- Troubleshooting bug that arise in application of these new alloys, ultimately improving product predictability.
When it comes to alloy development, phase diagrams have helped prevent overdesign for applications. This keeps price and procedure time down. They also assistance develop alternative alloys or same alloys with alternative alloying elements. It can help to reduce the need for using scarce, hazardous, or expensive alloying elements.
Performance-wise, phase diagrams help metallurgists sympathise which phases are thermodynamically stable, metastable, or unstable in the long run. Appropriate elements tin can then exist chosen for alloying to prevent machinery breakdown. Material for exhaust piping, for example, if non called properly, may lead to breakdown at higher temperatures.
The service life besides improves equally stage diagrams testify us how to solve problems such as intergranular corrosion, hot corrosion, and hydrogen damage.
Fe-Carbon Stage Diagram
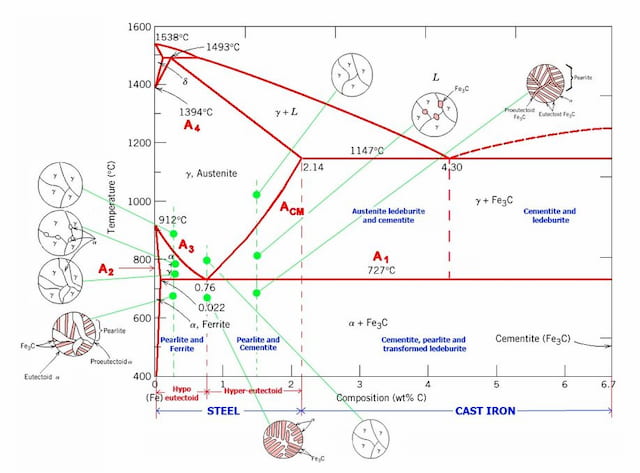
The iron-carbon phase diagram is widely used to understand the different phases of steel and cast atomic number 26. Both steel and cast iron are a mix of iron and carbon. Also, both alloys contain a small amount of trace elements.
The graph is quite complex but since we are limiting our exploration to Fe3C, nosotros will but be focusing upwardly to 6.67 weight percent of carbon.
This iron carbon phase diagram is plotted with the carbon concentrations by weight on the X-axis and the temperature calibration on the Y-axis.
The carbon in iron is an interstitial impurity. The alloy may class a face centred cubic (FCC) lattice or a trunk centred cubic (BCC) lattice. It will form a solid solution with α, γ, and δ phases of iron.
Types of Ferrous Alloys on the Phase Diagram
The weight percentage scale on the 10-axis of the iron carbon phase diagram goes from 0% up to half-dozen.67% Carbon. Up to a maximum carbon content of 0.008% weight of Carbon, the metal is simply chosen iron or pure iron. It exists in the α-ferrite form at room temperature.
From 0.008% upwardly to 2.14% carbon content, the iron carbon alloy is called steel. Inside this range, there are unlike grades of steel known equally low carbon steel (or mild steel), medium carbon steel, and high carbon steel.
When the carbon content increases beyond 2.fourteen%, we achieve the stage of bandage iron. Cast iron is very hard but its brittleness severely limits its applications and methods for forming.
Boundaries
Multiple lines can be seen in the diagram titled A1, A2, A3, A4, and ACM. The A in their proper name stands for the word 'arrest'. As the temperature of the metal increases or decreases, stage change occurs at these boundaries when the temperature reaches the value on the boundary.
Normally, when heating an alloy, its temperature increases. But along these lines (A1, A2, A3, A4, and ACM) the heating results in a realignment of the structure into a unlike stage and thus, the temperature stops increasing until the phase has changed completely. This is known as thermal arrest as the temperature stays constant.
Blend steel elements such as nickel, manganese, chromium, and molybdenum touch on the position of these boundaries on the phase diagram. The boundaries may shift in either direction depending on the element used. For example, in the iron carbon phase diagram, add-on of nickel lowers the A3 purlieus while the add-on of chromium raises it.
Eutectic Point
Eutectic signal is a point where multiple phases come across. For the iron-carbon alloy diagram, the eutectic indicate is where the lines A1, A3 and ACM meet. The formation of these points is coincidental.
At these points, eutectic reactions have identify where a liquid stage freezes into a mixture of two solid phases. This happens when cooling a liquid alloy of eutectic limerick all the way to its eutectic temperature.
The alloys formed at this betoken are known equally eutectic alloys. On the left and right side of this bespeak, alloys are known as hypoeutectic and hypereutectic alloys respectively ('hypo' in Greek ways less than, 'hyper' means greater than).
Phase Fields
The boundaries, intersecting each other, marking certain regions on the Fe3C diagram.
Within each region, a different stage or ii phases may exist together. At the boundary, the phase modify occurs. These regions are the phase fields.
They indicate the phases present for a certain composition and temperature of the alloy. Permit's acquire a little about the unlike phases of the iron-carbon blend.
Unlike Phases
α-ferrite
Existing at low temperatures and low carbon content, α-ferrite is a solid solution of carbon in BCC Fe. This phase is stable at room temperature. In the graph, information technology can be seen as a sliver on the left border with Y-axis on the left side and A2 on the right. This stage is magnetic below 768°C.
It has a maximum carbon content of 0.022 % and it will transform to γ-austenite at 912°C as shown in the graph.
γ-austenite
This phase is a solid solution of carbon in FCC Iron with a maximum solubility of 2.14% C. On further heating, it converts into BCC δ-ferrite at 1395°C. γ-austenite is unstable at temperatures below eutectic temperature (727°C) unless cooled rapidly. This phase is non-magnetic.
δ-ferrite
This stage has a similar structure as that of α-ferrite but exists but at high temperatures. The phase can exist spotted at the top left corner in the graph. It has a melting point of 1538°C.
Fe3C or cementite
Cementite is a metastable phase of this blend with a stock-still limerick of Fe3C. Information technology decomposes extremely slowly at room temperature into Iron and carbon (graphite).
This decomposition time is long and it will take much longer than the service life of the application at room temperature. Some other factors (loftier temperatures and addition of certain alloying elements for instance) can affect this decomposition equally they promote graphite germination.
Cementite is hard and breakable which makes it suitable for strengthening steels. Its mechanical backdrop are a function of its microstructure, which depends upon how it is mixed with ferrite.
Atomic number 26-C liquid solution
Marked on the diagram every bit '50', it tin be seen in the upper region in the diagram. As the proper name suggests, information technology is a liquid solution of carbon in fe. As we know that δ-ferrite melts at 1538°C, it is evident that melting temperature of iron decreases with increasing carbon content.
Source: https://fractory.com/iron-carbon-phase-diagram/
Posted by: wilsonevembee.blogspot.com



0 Response to "How To Draw Microstructure From Phase Diagram"
Post a Comment